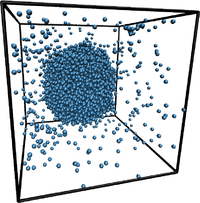
 Metastable phases decay by rare statistical fluctuations, termed "nucleation", where a "critical cluster" (i.e., a nanoscopic aggregate of the new phase having the minimal size to be able to grow) forms. Such critical clusters may form in the bulk ("homogeneous nucleation"), which facilitate this cluster formation by reducing the free energy barrier that needs to be overcome. Specialized computer simulation methods are developed to estimate the surface excess free energies, that control these processes, and related quantities (such as contact angles, line tension, Tolman length). This research is carried out in the framework of the priority program SPP 1296 "Heterogeneous Nucleation and Structure Formation" of the German Research Foundation (DFG), and we collaborate with J. Horbach (DLR Cologne) and S. Egelhaaf and H. Löwen (University of Düsseldorf), where complementary studies are done, as well as with S. K. Das (Bangalore) and S. Puri (New Delhi, India). For more information, please contact Kurt Binder and/or Peter Virnau. Metastable phases decay by rare statistical fluctuations, termed "nucleation", where a "critical cluster" (i.e., a nanoscopic aggregate of the new phase having the minimal size to be able to grow) forms. Such critical clusters may form in the bulk ("homogeneous nucleation"), which facilitate this cluster formation by reducing the free energy barrier that needs to be overcome. Specialized computer simulation methods are developed to estimate the surface excess free energies, that control these processes, and related quantities (such as contact angles, line tension, Tolman length). This research is carried out in the framework of the priority program SPP 1296 "Heterogeneous Nucleation and Structure Formation" of the German Research Foundation (DFG), and we collaborate with J. Horbach (DLR Cologne) and S. Egelhaaf and H. Löwen (University of Düsseldorf), where complementary studies are done, as well as with S. K. Das (Bangalore) and S. Puri (New Delhi, India). For more information, please contact Kurt Binder and/or Peter Virnau.
|